MIDLOTHIAN, Va – Pulmotect reported positive topline results from their randomized, placebo-controlled Phase-2 trial of PUL-042 against COVID-19. Patients treated with inhaled PUL-042 had a statistically significant reduction in the time to improvement of the combined respiratory symptoms of cough and shortness of breath.
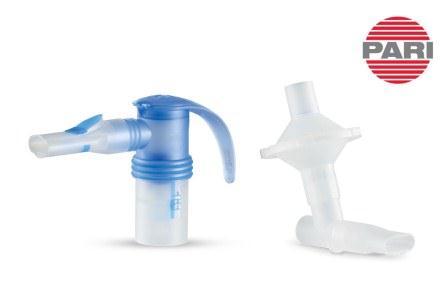
Pulmotect’s PUL-042, an inhaled immunomodulatory agent to prevent and treat coronavirus infections was delivered by PARI’s LC Sprint Reusable Nebulizer. PARI Nebulizers and systems are the most widely used nebulizers by pharmaceutical companies developing new nebulized medications for delivery to the lungs.
“We have demonstrated PUL-042’s unique ability to stimulate the immune system in the lungs to protect against a wide range of pathogens in multiple animal models,” said Colin Broom, MD, Chief Executive Officer of Pulmotect. “We are delighted to follow up with evidence of clinical activity from our double blind, randomized, placebo-controlled Phase 2 clinical trial in patients with early COVID-19. PUL-042 was administered by nebulization on Day 1, Day 3, and Day 6 of the trial with patient follow up to Day 29.
“PARI’s LC Sprint Nebulizer equipped with a PARI Filter Valve Set to reduce potential fugitive aerosol emission and the Vios PRO compressor (now PRONEB Max) exclusively delivered PUL-042 to subjects in the Phase 2 study. We want to congratulate the Pulmotect team on the positive topline results of the PUL-042 program which could provide a significant benefit to those with COVID-19.”, said Lisa Cambridge, MSHS, RRT, Director of Medical Science at PRE Holding, Inc. “It is another great example of PARI’s nebulizers being used successfully in new clinical trials for respiratory indications, including COVID-19”.